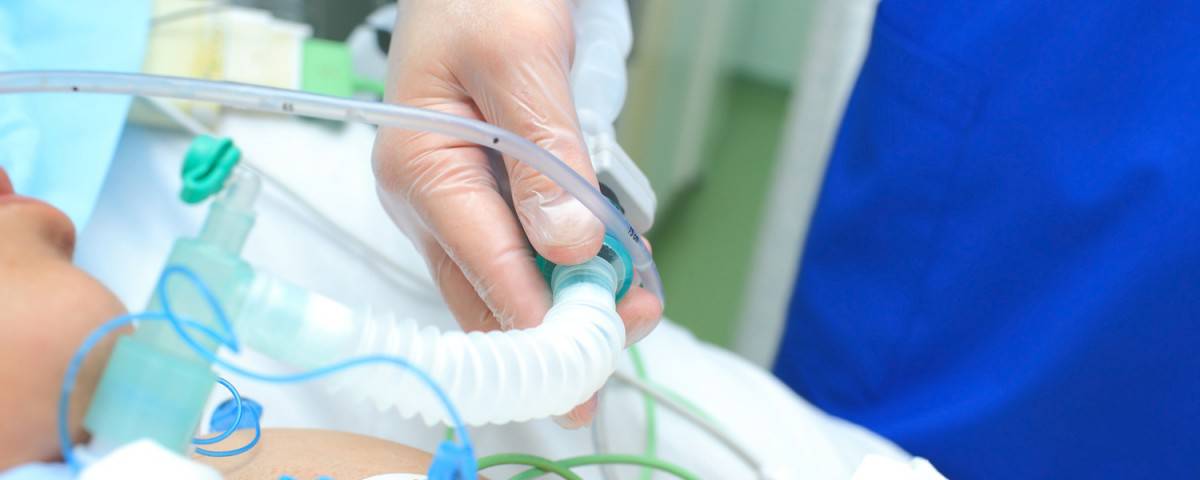
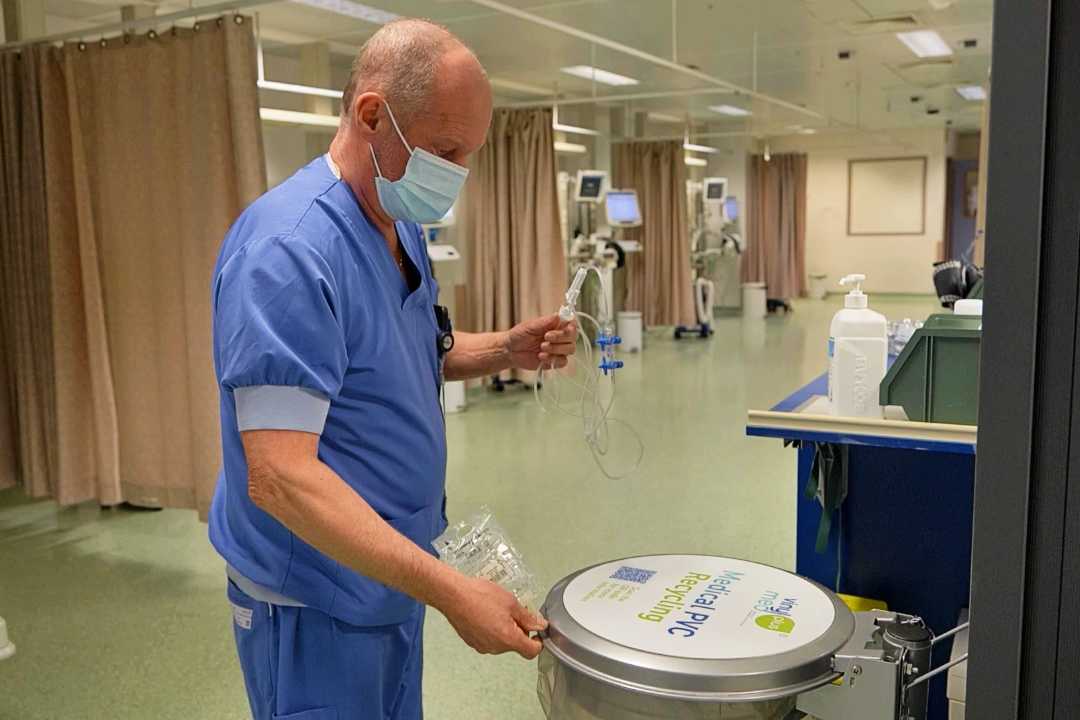
Recycling of plastics waste from healthcare will increase
16/07/2013
International conference on Alternatives to Classified Phthalates in PVC Medical Devices
26/02/2014The PVCMed Alliance issues the following statement in response to the article “Broad-based assault on phthalates coming in the EU“ published by PlasticsToday.
The PVCMed Alliance acknowledges the fact that some phthalates used as plasticisers in a number of medical devices are currently under growing scrutiny by regulatory and medical authorities. PVCMed does not favour any specific plasticisers used in the medical devices. Rather, it encourages the value chain to commit to innovation, continuous improvement of safety, performance and cost efficiency in order to continue developing innovative products for all healthcare applications.
However, we fear that the article above can create confusion by over generalising to the entire medical applications made of PVC. As a matter of fact, the article underestimates the various qualities of PVC that have not been equalled by any other material for medical applications. PVC is to date the only material that is able to guarantee the quality and performance demanded by medical professionals and regulatory bodies, while still remaining highly cost-efficient.
The PVCMed Alliance reiterates the willingness of the industry to innovate and to continuously improve it products. As a matter of fact, alternative plasticisers that perform their technological function with PVC as well as phthalate plasticisers have been developed and are increasingly being used in a wide array of medical applications allowing medical equipment purchasers to benefit from PVC’s unique properties for patient comfort, economic affordability and hospital hygiene.