Plastics – more than straws and cotton buds
11/04/2019
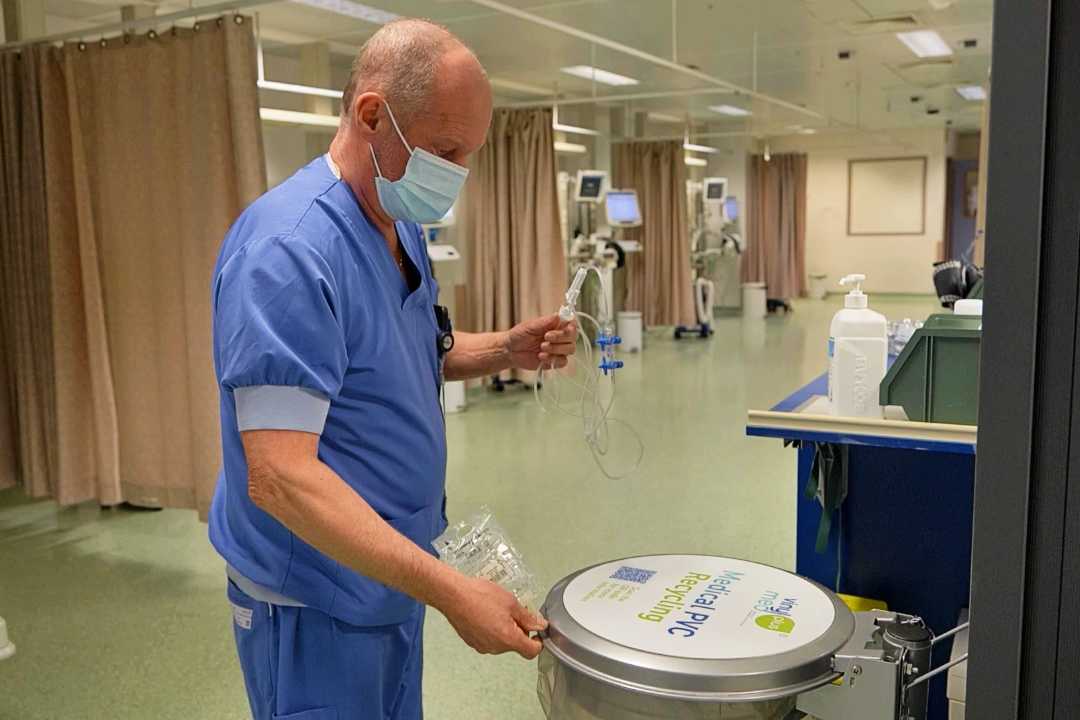
Busy nurses drive sustainable development in healthcare
27/06/2019In 2017 the new EU medical device and in vitro diagnostic regulations (MDR/IVDR) entered into force. As the three-year transition period for the MDR is about to end (spring 2020), the European Commission has issued a fact sheet for healthcare professionals and health institutions to explain the impact of the new regulations.
The fact sheet acknowledges that the new regulations can “have consequences for the availability of medical devices for health institutions. For instance, manufacturers may choose to stop the production of certain medical devices. Furthermore, if certain medical devices do not get their certificates on time these products may become temporarily unavailable.”
In other news, the Scientific Committee on Health, Environmental and Emerging Risks (SCHEER) is expected to publish its final guidelines on the benefit-risk assessment of the presence of phthalates in certain medical devices covering phthalates which are carcinogenic, mutagenic, toxic to reproduction (CMR) or have endocrine-disrupting properties very soon. Public consultation ended 29 April 2019 and a hearing was held on 4 April 2019.
Find here a European Commission fact sheet for medical device manufacturers.